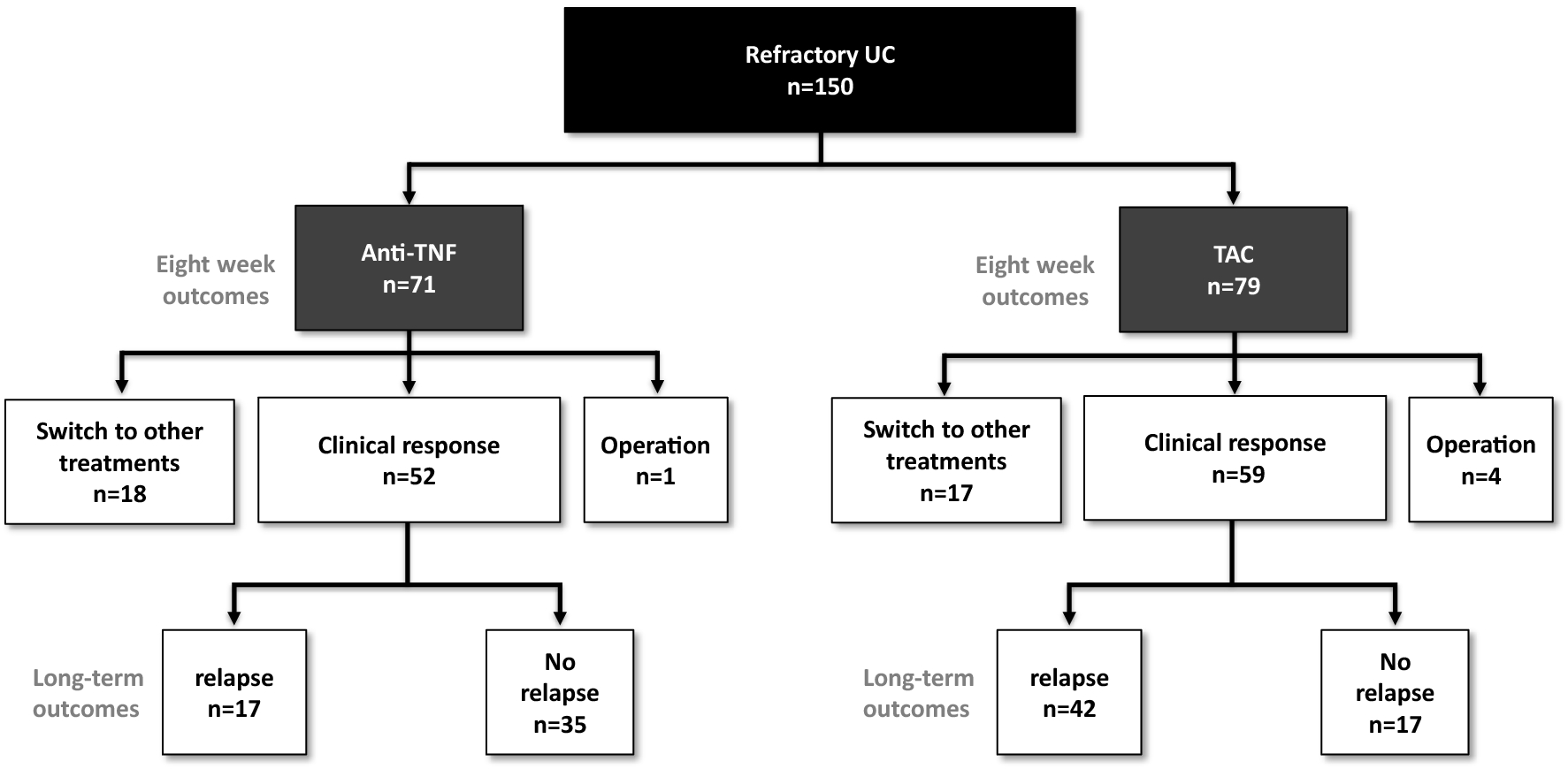
Comparative efficacy of antitumor necrosis factor agents and tacrolimus in naïve steroid-refractory ulcerative colitis patients | Scientific Reports
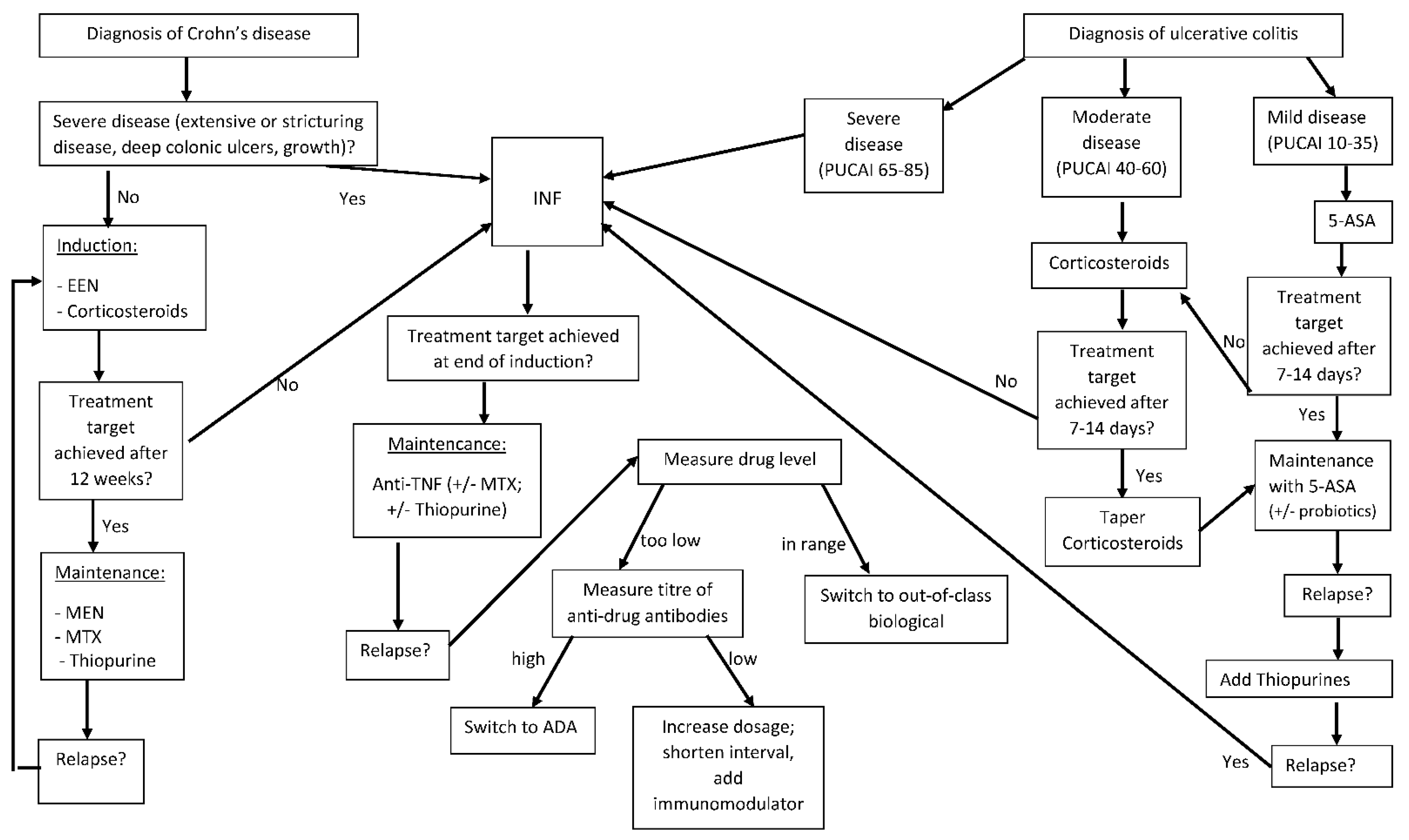
Children | Free Full-Text | Therapy Strategies for Children Suffering from Inflammatory Bowel Disease (IBD)—A Narrative Review | HTML
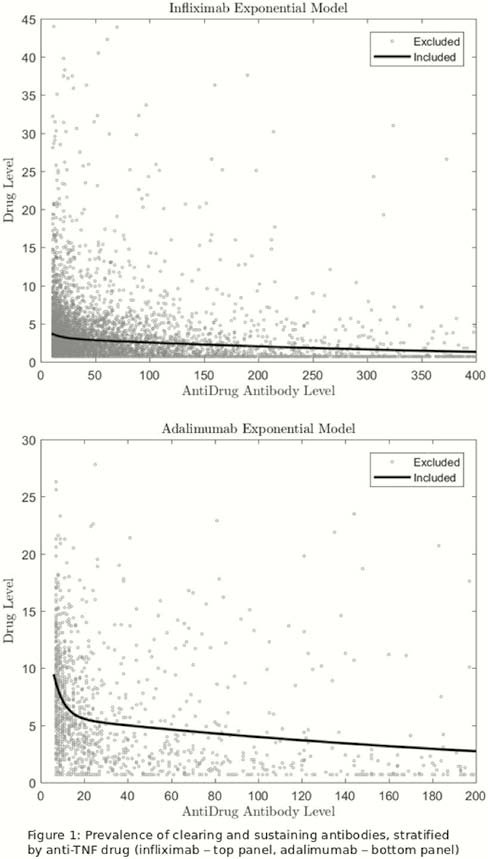
European Crohn´s and Colitis Organisation - ECCO - OP21 Positivity thresholds of total infliximab and adalimumab anti-drug antibody assay: The prevalence of clearing and transient anti-drug antibodies in a national therapeutic drug
Third European Evidence-based Consensus on Diagnosis and Management of Ulcerative Colitis. Part 1: Definitions, Diagnosis, Extra

Rational Combination Therapy to Overcome the Plateau of Drug Efficacy in Inflammatory Bowel Disease - Gastroenterology

Histologic Outcomes With Vedolizumab Versus Adalimumab in Ulcerative Colitis: Results From An Efficacy and Safety Study of Vedolizumab Intravenous Compared to Adalimumab Subcutaneous in Participants With Ulcerative Colitis (VARSITY) - Gastroenterology

Proactive therapeutic drug monitoring of adalimumab for pediatric Crohn's disease patients: A cost‐effectiveness analysis - Yao - 2021 - Journal of Gastroenterology and Hepatology - Wiley Online Library
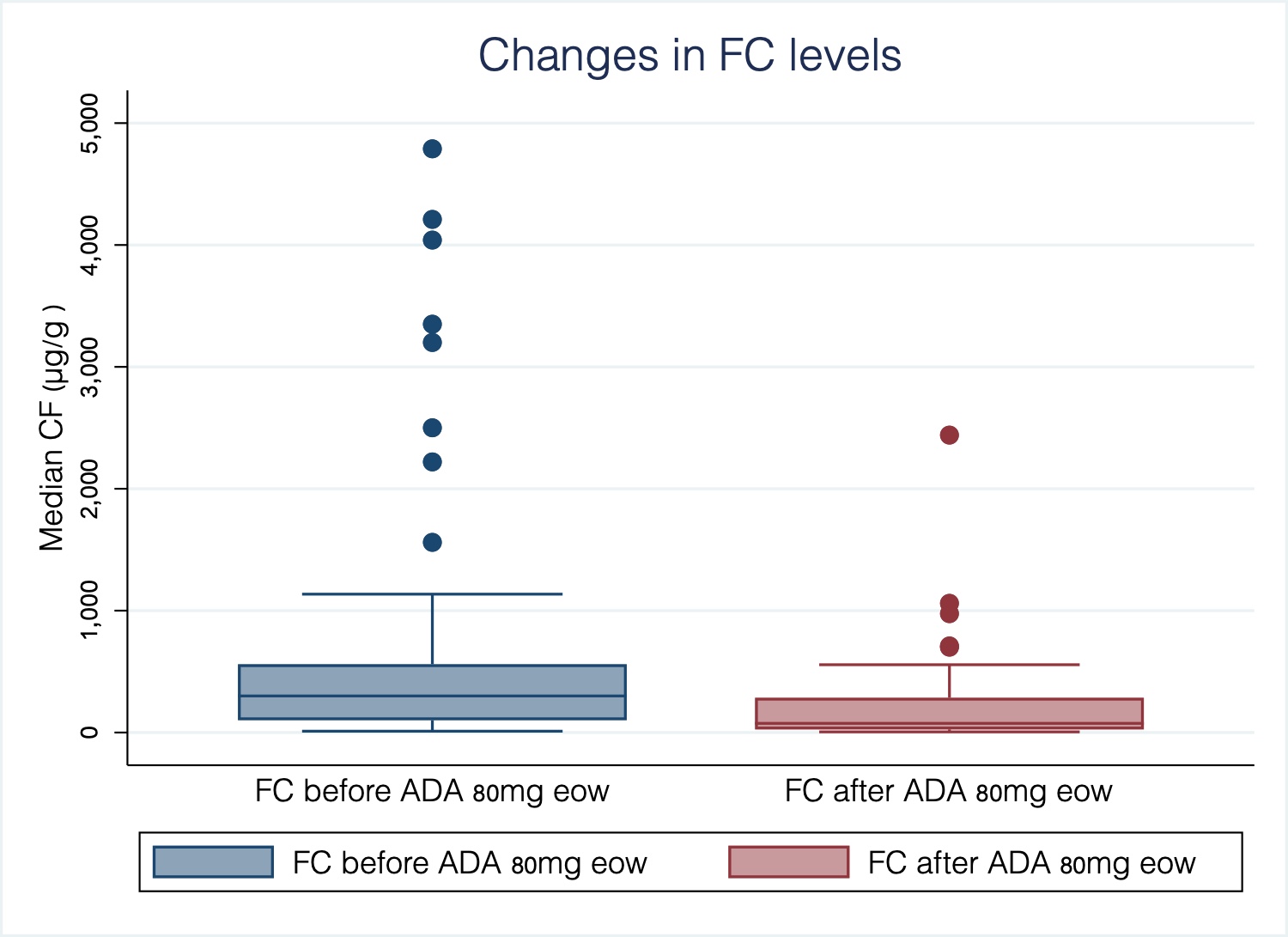
European Crohn´s and Colitis Organisation - ECCO - P533 Adalimumab 80mg every other week in inflammatory bowel disease: Treatment intensification outcomes in real life clinical practice

The Toronto Consensus Statements for the Management of Inflammatory Bowel Disease in Pregnancy - Gastroenterology
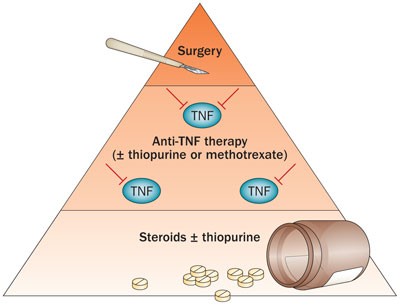
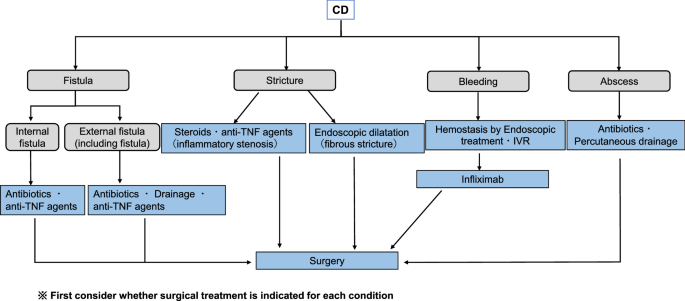
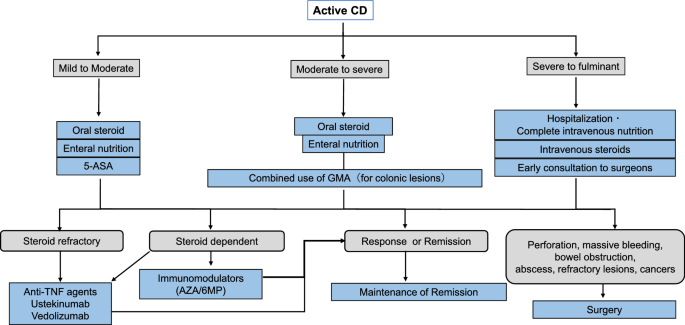
3rd European Evidence-based Consensus on the Diagnosis and Management of Crohn's Disease 2016: Part 1: Diagnosis and Medical M

European Crohn´s and Colitis Organisation - ECCO - DOP62 Immunogenicity to second anti-TNF therapy (IMSAT): Implications for sequencing of biologic therapy

Immunomodulator Withdrawal From Anti-TNF Therapy Is Not Associated With Loss of Response in Inflammatory Bowel Disease - Clinical Gastroenterology and Hepatology

European Crohn´s and Colitis Organisation - ECCO - DOP62 Immunogenicity to second anti-TNF therapy (IMSAT): Implications for sequencing of biologic therapy

Efficacy and safety of adalimumab in paediatric patients with moderate-to-severe ulcerative colitis (ENVISION I): a randomised, controlled, phase 3 study - The Lancet Gastroenterology & Hepatology

PDF) Adalimumab vs Azathioprine in the Prevention of Postoperative Crohn's Disease Recurrence. A GETECCU Randomised Trial
![PDF] Report of the ECCO pathogenesis workshop on anti-TNF therapy failures in inflammatory bowel diseases: definitions, frequency and pharmacological aspects. | Semantic Scholar PDF] Report of the ECCO pathogenesis workshop on anti-TNF therapy failures in inflammatory bowel diseases: definitions, frequency and pharmacological aspects. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/434bc5078bbc62a1a4782f27592fcf28f239679e/8-Table2-1.png)
PDF] Report of the ECCO pathogenesis workshop on anti-TNF therapy failures in inflammatory bowel diseases: definitions, frequency and pharmacological aspects. | Semantic Scholar
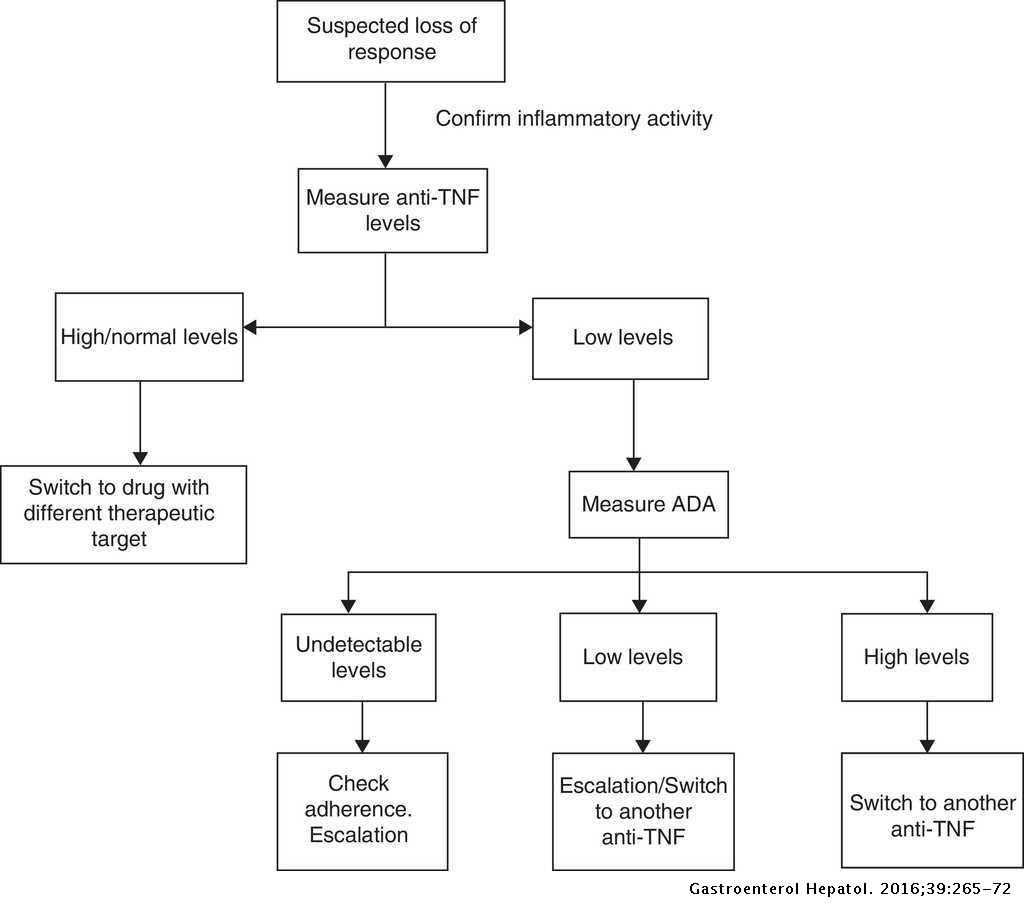
Drugs and anti-drug antibody levels in the management of patients with inflammatory bowel disease | Gastroenterología y Hepatología (English Edition)

Proactive Monitoring of Adalimumab Trough Concentration Associated With Increased Clinical Remission in Children With Crohn's Disease Compared With Reactive Monitoring - Gastroenterology