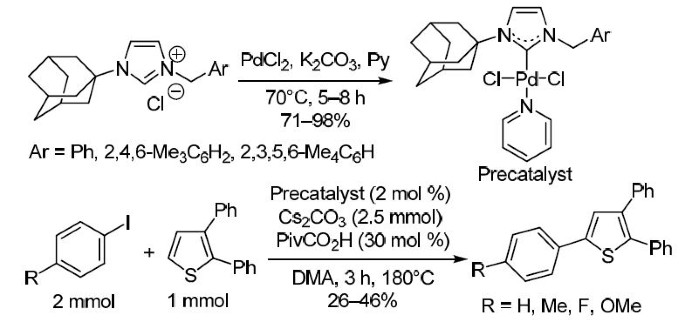
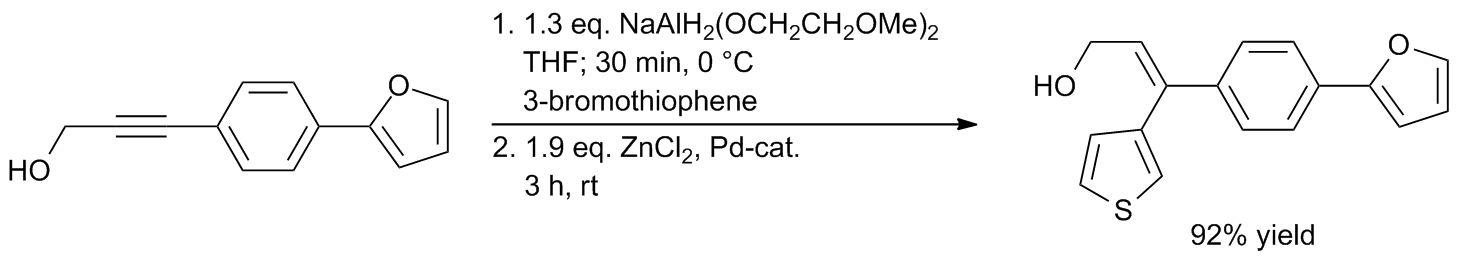
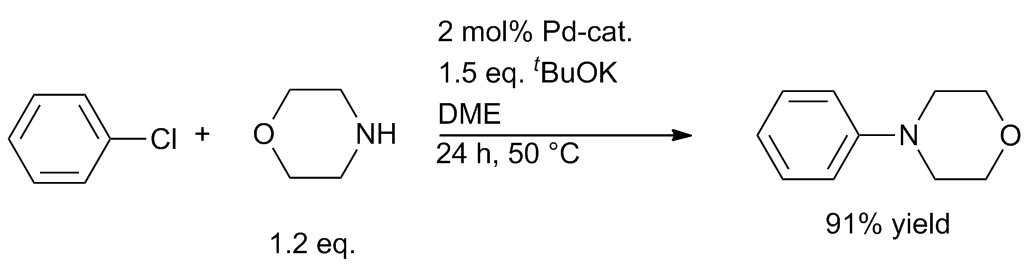
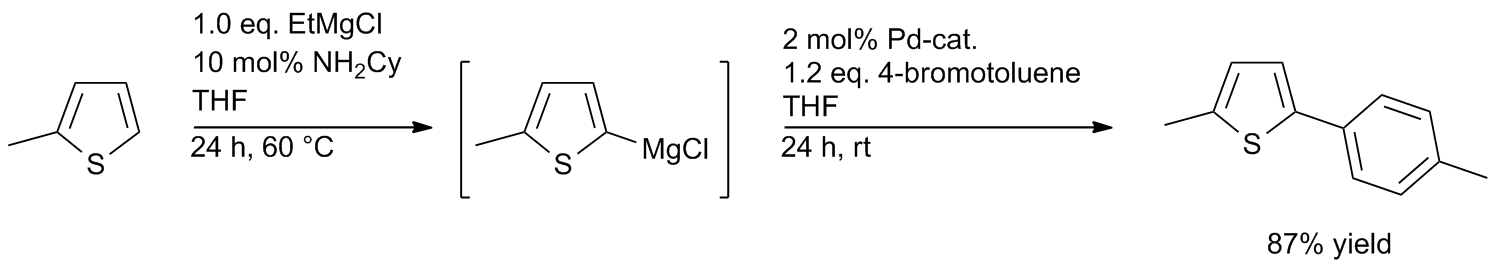
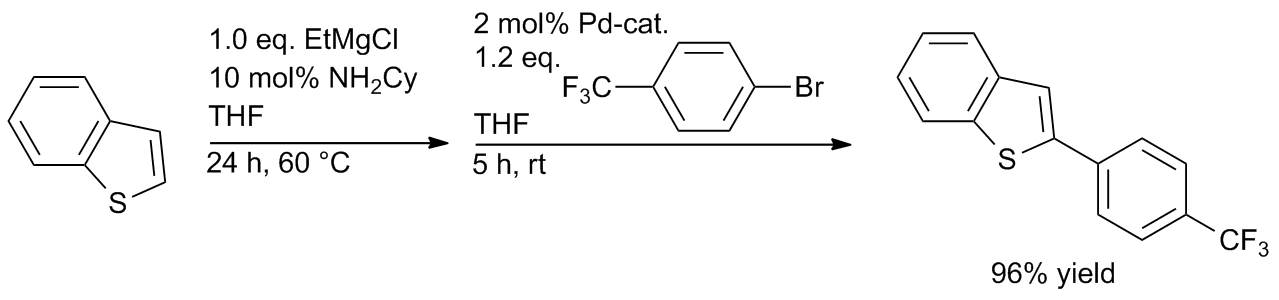
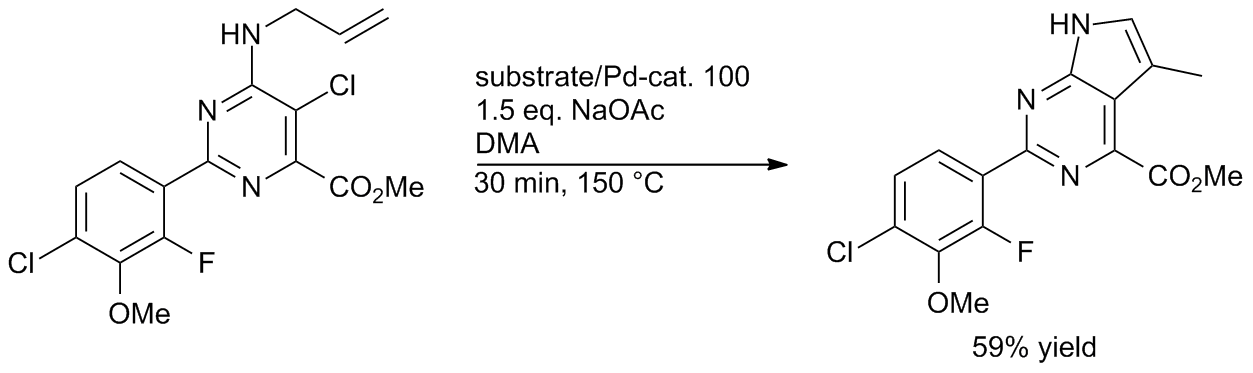
Well‐defined PEPPSI‐themed palladium–NHC complexes: synthesis, and catalytic application in the direct arylation of heteroarenes - Kaloğlu - 2020 - Applied Organometallic Chemistry - Wiley Online Library
Synergy between supported ionic liquid-like phases and immobilized palladium N-heterocyclic carbene–phosphine complexes for th

Investigation of activity, stability, and degradation mechanism of surface-supported Pd-PEPPSI complexes for Suzuki-Miyaura coupling - ScienceDirect

Room‐Temperature Amination of Deactivated Aniline and Aryl Halide Partners with Carbonate Base Using a Pd‐PEPPSI‐IPentCl‐ o‐Picoline Catalyst - Pompeo - 2014 - Angewandte Chemie International Edition - Wiley Online Library

Synthesis, characterization and catalytic activity of PEPPSI-type palladium–NHC complexes - ScienceDirect

Cycloheptyl substituted N-heterocyclic carbene PEPPSI-type palladium complexes with different N-coordinated ligands: Involvement in Suzuki-Miyaura reaction - ScienceDirect
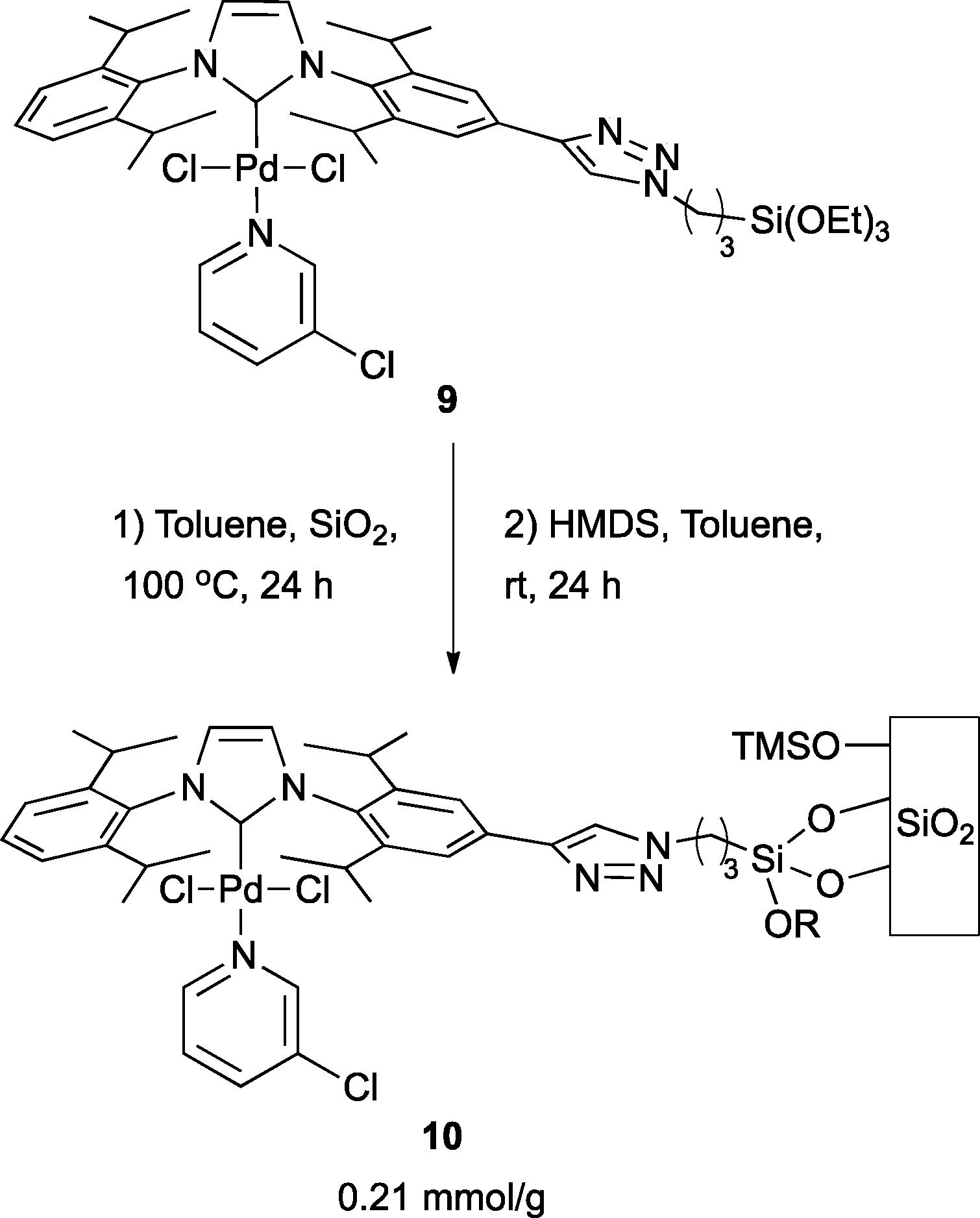
Continuous flow Negishi cross-couplings employing silica-supported Pd-PEPPSI – IPr precatalyst - Catalysis Science & Technology (RSC Publishing) DOI:10.1039/C6CY00331A
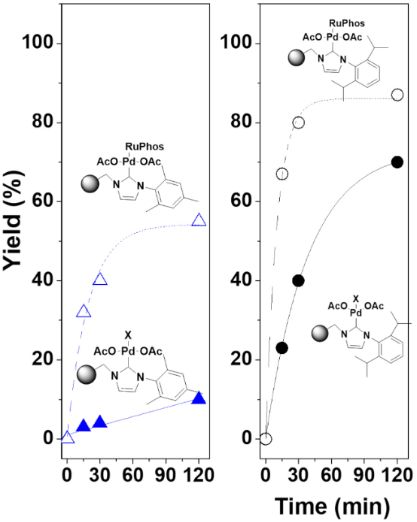
Synergy between supported ionic liquid-like phases and immobilized palladium N-heterocyclic carbene–phosphine complexes for the Negishi reaction under flow conditions

Synthesis of bridged palladium-PEPPSI complexes and catalytic studies in C–C cross-coupling reactions - ScienceDirect

Pd‐PEPPSI‐IPent‐SiO2: A Supported Catalyst for Challenging Negishi Coupling Reactions in Flow - Price - 2017 - Angewandte Chemie International Edition - Wiley Online Library

PDF) Kinetic Study of the Herrmann–Beller Palladacycle-Catalyzed Suzuki–Miyaura Coupling of 4-Iodoacetophenone and Phenylboronic Acid

Phase‐Separable Polyisobutylene Palladium‐PEPPSI Precatalysts: Synthesis and Application in Buchwald–Hartwig Amination - Balogh - 2017 - Macromolecular Rapid Communications - Wiley Online Library
Polytriarylamines Containing Fused Ring and Heterocyclic Structures Prepared Using -Heterocyclic Carbene Complexes of Palladium
![Catalysts | Free Full-Text | Palladium PEPPSI-IPr Complex Supported on a Calix[8]arene: A New Catalyst for Efficient Suzuki–Miyaura Coupling of Aryl Chlorides | HTML Catalysts | Free Full-Text | Palladium PEPPSI-IPr Complex Supported on a Calix[8]arene: A New Catalyst for Efficient Suzuki–Miyaura Coupling of Aryl Chlorides | HTML](https://www.mdpi.com/catalysts/catalysts-10-01081/article_deploy/html/images/catalysts-10-01081-g001.png)
Catalysts | Free Full-Text | Palladium PEPPSI-IPr Complex Supported on a Calix[8]arene: A New Catalyst for Efficient Suzuki–Miyaura Coupling of Aryl Chlorides | HTML
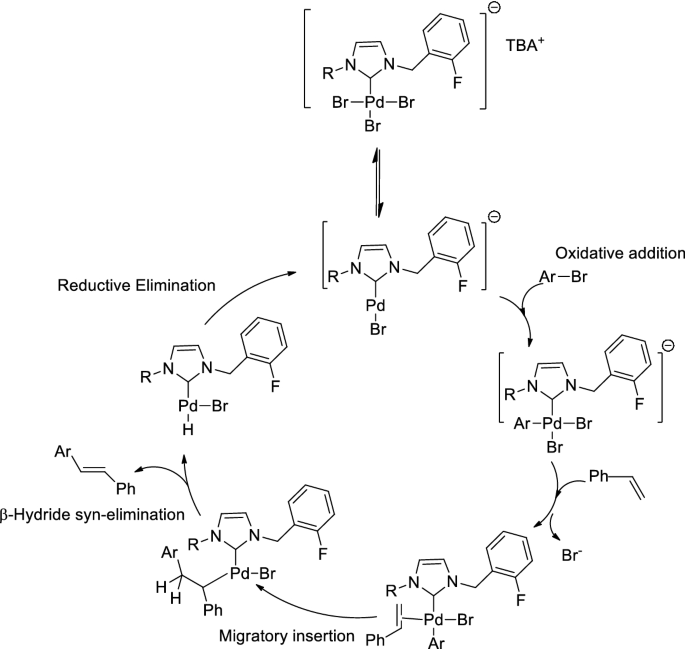
A new PEPPSI type N-heterocyclic carbene palladium(II) complex and its efficiency as a catalyst for Mizoroki-Heck cross-coupling reactions in water | SpringerLink

Sulfination by Using Pd‐PEPPSI Complexes: Studies into Precatalyst Activation, Cationic and Solvent Effects and the Role of Butoxide Base - Sayah - 2013 - Chemistry – A European Journal - Wiley Online Library

Benzimidazole bearing Pd–PEPPSI complexes catalyzed direct C2‐arylation/heteroarylation of N‐substituted benzimidazoles - Gokanapalli - - Applied Organometallic Chemistry - Wiley Online Library